OPENS ON 24/04/2026
atomicsciencemadeasy
-
Welcome to Hilder's School of Analytical Chemistry
Analytical Chemistry is a foundational course that focuses on the principles, techniques, and applications used to identify, separate, and quantify chemical substances. This course introduces students to both classical (wet) analytical methods and modern instrumental techniques essential for chemical analysis in laboratories, industry, medicine, environmental science, and research.
Vision
To be a leading center of excellence in Analytical Chemistry education, research, and innovation, producing skilled professionals who contribute to scientific advancement, sustainable development, and global problem-solving.
Mission
To provide high-quality education and practical training in Analytical Chemistry through innovative teaching, rigorous laboratory experience, and impactful research.
We are committed to developing competent, ethical, and analytical scientists capable of delivering accurate chemical analysis, advancing scientific knowledge, and addressing real-world environmental, industrial, and health challenges.Core Values
1. Scientific Integrity
We uphold honesty, accuracy, and ethical behavior in all laboratory work, research activities, and academic engagements.
2. Excellence
We strive for high standards in teaching, learning, research, and analytical performance.
3. Innovation
We encourage creativity and the adoption of modern analytical techniques and technologies that enhance scientific discovery.
4. Precision and Accuracy
We value careful measurement, proper method validation, and data reliability, which form the foundation of analytical chemistry.
5. Safety and Responsibility
We prioritize safe laboratory practices, proper chemical handling, and environmental protection.
6. Collaboration
We promote teamwork, interdisciplinary partnerships, and industry linkages to solve scientific and societal challenges.
(You can edit or remove this text)
Site announcements
Skip course categories
Course categories
Skip available courses
Available courses
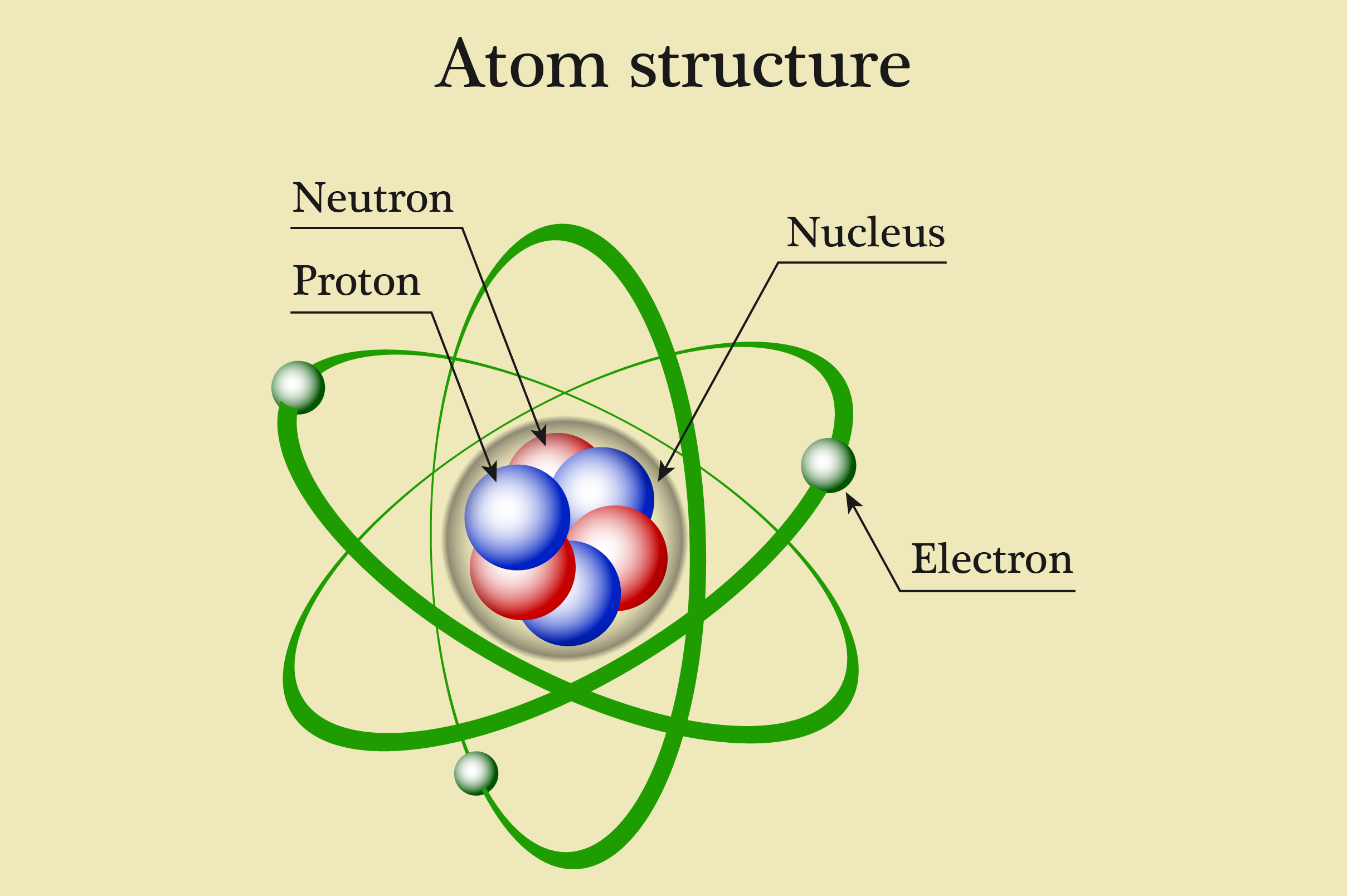
Course Description: Atomic Structure
This concise course provides a fundamental understanding of the atom, the basic building block of matter. Students will explore the historical development of atomic theory, from early philosophical ideas to modern quantum mechanical models.
Key Topics Covered
The course focuses on the internal organization and properties of the atom, including:
Subatomic Particles: Identification and characteristics of protons (positive charge, found in the nucleus), neutrons (no charge, found in the nucleus), and electrons (negative charge, orbiting the nucleus in shells/orbitals).
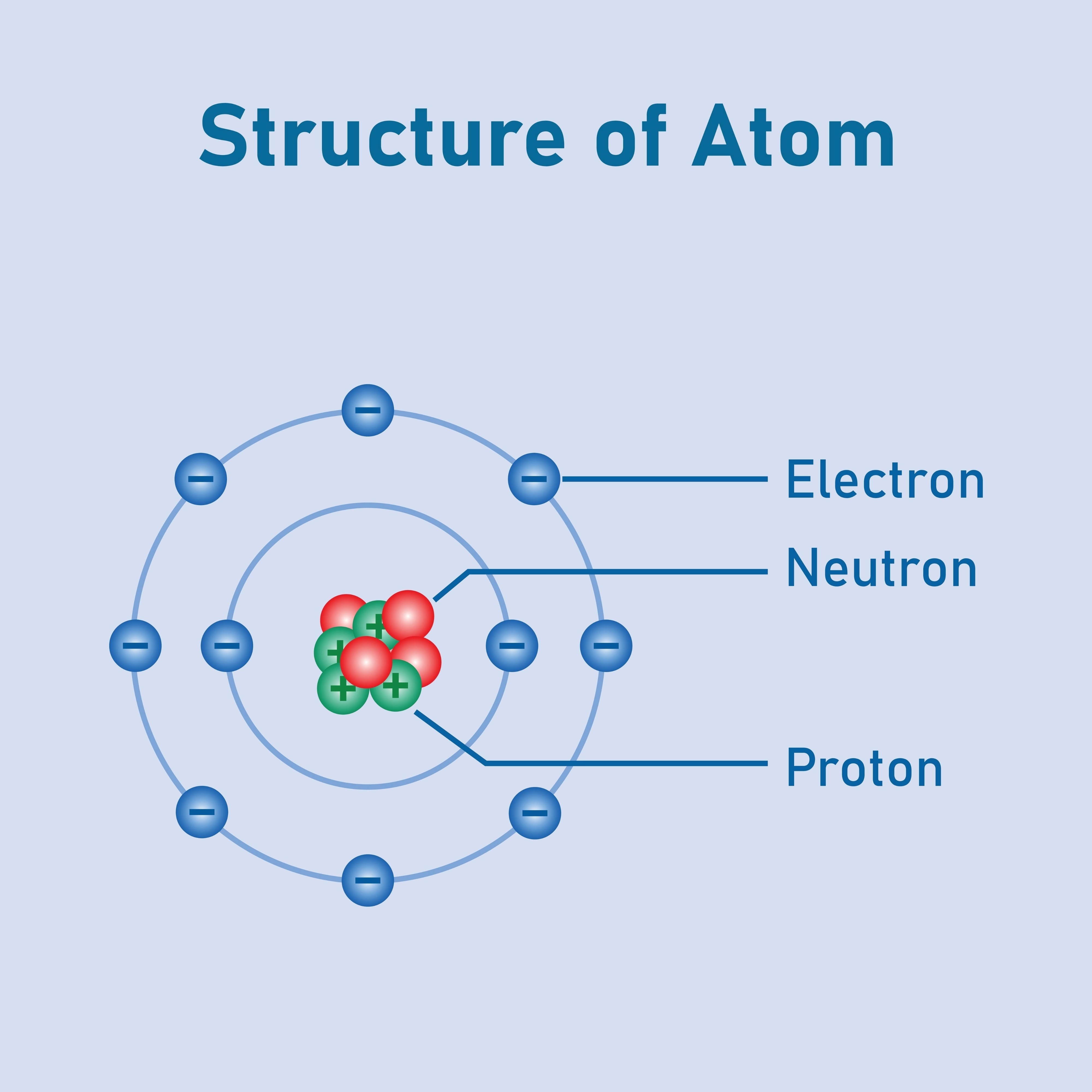
Atomic Definitions: Understanding key terms like Atomic Number (Z) (number of protons), Mass Number (A) (protons + neutrons), and the concept of Isotopes (atoms of the same element with different numbers of neutrons).
Historical Models: A review of influential atomic models, such as Dalton's, Thomson's (Plum Pudding), Rutherford's (Nuclear Model), and Bohr's model, highlighting how scientific understanding evolved.
Electron Configuration & Quantum Mechanics: Introduction to the modern Quantum Mechanical Model, including electron shells (energy levels), subshells (s, p, d, f), orbitals, and the rules governing how electrons fill these energy levels (Aufbau principle, Hund's rule, Pauli exclusion principle).
Periodic Trends: Analyzing how atomic structure dictates the organization of the Periodic Table and influences periodic properties like atomic radius, ionization energy, and electronegativity
Ions and Stability: Explaining how atoms gain or lose electrons to form ions and achieve stable electronic configurations, which is foundational to understanding chemical bonding.
- Teacher: Hilder Kosgei
ATOMIC STRUCTURE 1.
- Teacher: Hilder Kosgei
Skip courses